 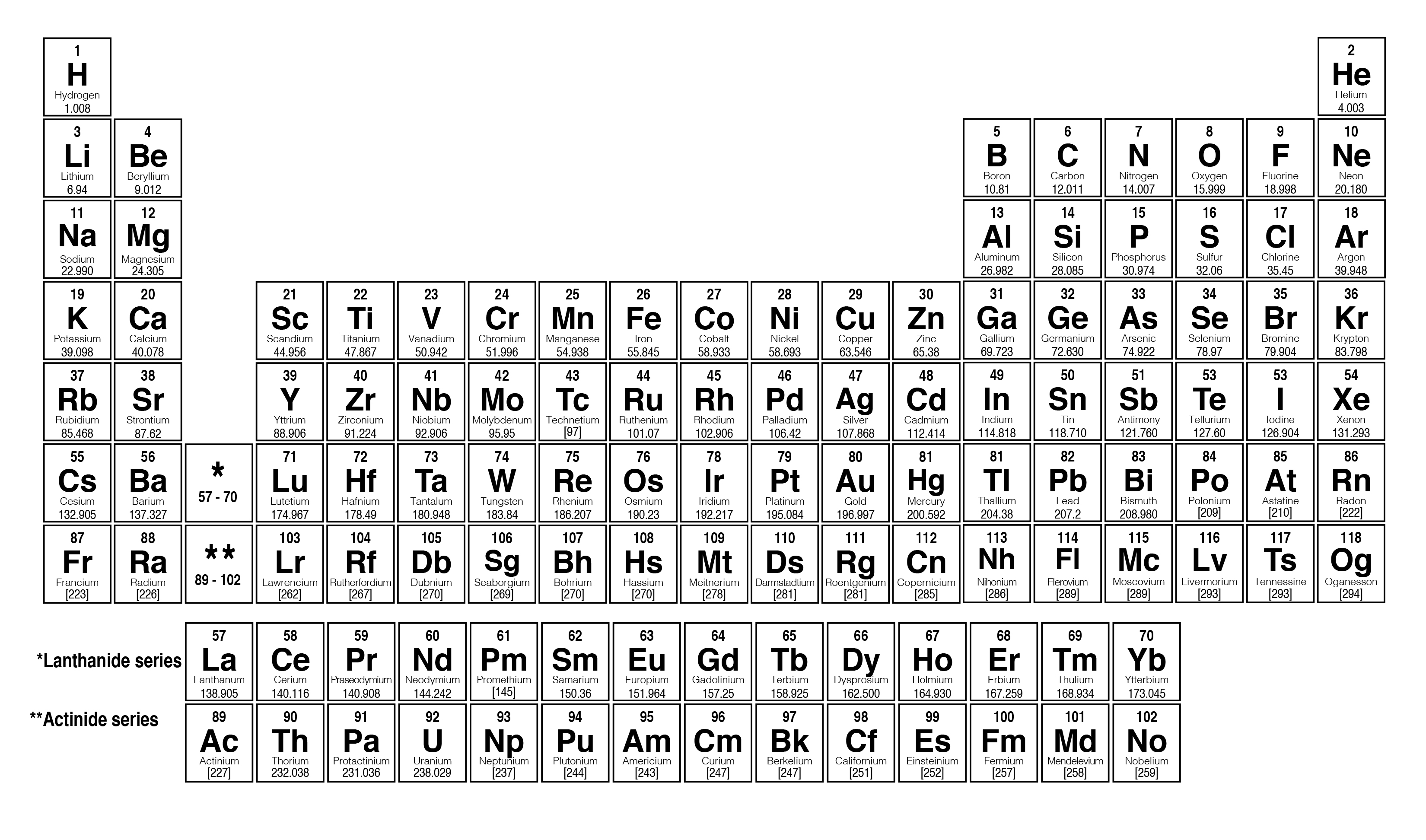
Its atomic number is 2, which is not always included in the notation because He is defined by the atomic number 2. Each isotope has a certain percentage abundance found in nature, and these are added and averaged to obtain the average mass number.įor example, 4He has a mass number of 4. The mass number listed is the average mass of all of the element's isotopes. If you compare these values with those given for some of the. The periodic table lists the atomic masses of all the elements. On the periodic table, the mass number is usually located below the element symbol. C Give the symbol of each isotope with the mass number as the superscript and the number of protons as the subscript, both written to the left of the symbol of the element. Key Takeaways: Number of Protons, Neutrons, and Electrons Atoms are made of protons, neutrons, and electrons. The symbol is a one- or two-letter abbreviation. Follow these simple steps to find the number of protons, neutrons, and electrons for an atom of any element. Element identification can be done using the atomic number, element name, or element symbol. There may be a few more or a few less neutrons, and so the mass is increased or decreased. Updated on JThe three parts of an atom are positive-charged protons, negative-charged electrons, and neutral neutrons. The oxidation number of an atom is indicated by a superscript in roman numerals on the right side of the chemical symbol. The two added together results in the mass number:Įlements can also have isotopes with the same atomic number, but different numbers of neutrons. 
Each proton and neutron's mass is approximately one atomic mass unit (AMU). Because an electron has negligible mass relative to that of a proton or a neutron, the mass number is calculated by the sum of the number of protons and neutrons. The mass of an atom is mostly localized to the nucleus. Examples as seen on Periodic Table Atomic NumberĪverage Atomic Mass of all Element's Isotopes Copper has the atomic number of 29 for its 29 protons. For example, hydrogen has one proton and one electron, so it has an atomic number of 1. Click on the group number for the Halogen family at the top of the table. The atomic number is usually located above the element symbol. The heaviest element in nature is uranium. On the periodic table, the elements are arranged in the order of atomic number across a period. Every element has a different atomic number, ranging from 1 to over 100. O 2- still has an atomic number of 8, corresponding to the 8 protons, but it has 10 electrons. The RutherfordBohr model of the hydrogen atom ( Z 1) or a hydrogen-like ion ( Z > 1 ). Atomic number is the number of protons, and therefore also the total positive charge, in the atomic nucleus. In the element symbol, the charge goes on the right side of the element. An explanation of the superscripts and subscripts seen in atomic number notation. If it is charged, there may be more protons than electrons or vice versa, but the atomic number remains the same. If an atom is neutrally charged, it has the same number of protons and electrons. \)Įvery element has a defining atomic number, with the symbol "Z". 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
